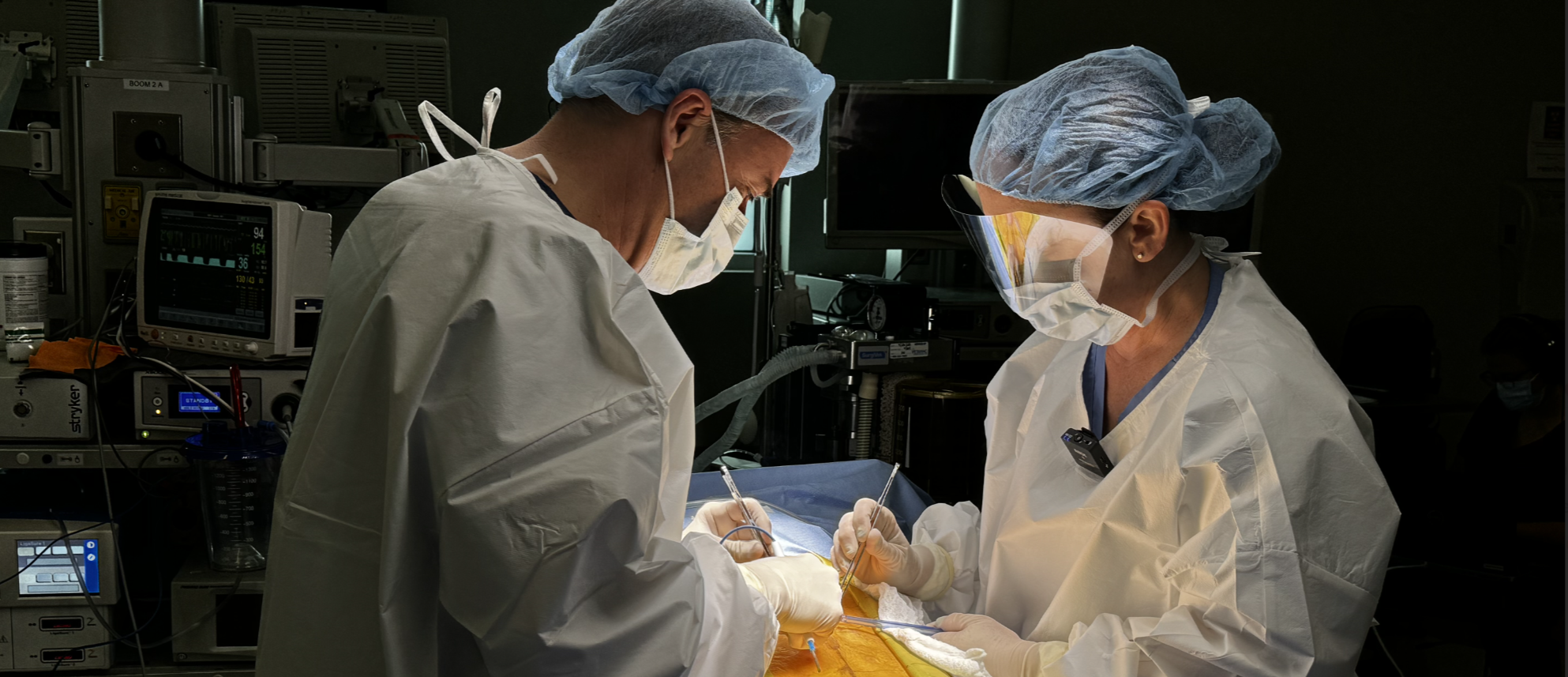
Like Coleridge’s Ancient Mariner, today’s surgeon often feels adrift on a sea of data — data everywhere, but not a byte to decide.
Large language models have proven valuable for novices: medical students, trainees, and clinicians looking to rapidly synthesize current guidelines. But for experienced surgeons making complex intraoperative decisions, these tools often fall short. They lack the nuance of years of clinical judgment and can occasionally miss the mark entirely.
So the question becomes: how do we turn the ocean of clinical data surrounding modern healthcare into something surgeons can actually use to improve outcomes?
Consider spine surgery. Today, many surgical decisions—instrumentation choices, level selection, alignment strategy—still rely primarily on the surgeon’s personal experience, supplemented by literature and peer discussion. That individual “database” is powerful but limited.
The cost of those limitations is real. In the U.S. alone, failed spine surgery accounts for roughly $5B in annual healthcare spend and according to one report, as much as $20B, often driven by incomplete or poorly synthesized information at the time of decision-making.
Innovations are emerging. Systems like advanced surgical navigation provide imaging-enhanced “eyes” to guide hardware placement, while new platforms are beginning to leverage aggregated datasets to support planning. These are important steps forward.
But most current systems share a constraint: their intelligence is limited by the datasets they were trained on. Further, the inherent segmentation of knowledge in for example imaging data, may not provide the whole picture.
Now imagine a different model.
What if the “training dataset” wasn’t a purchased proprietary database—but the entire clinical data ecosystem of a health system itself?
Within most hospital systems already exists a decade or more of rich clinical information:
- patient demographics
- imaging and operative data
- treatment decisions across hundreds of physicians
- long-term outcomes
- and often cost and resource utilization
In addition, by enabling multi-variable consideration within a single model, we would be able to provide the best possible care, more closely tailored to an individual patient’s needs.
In that environment, surgical planning tools no longer rely solely on static historical training sets and subsets of clinical variables. Instead, they draw from living clinical intelligence, helping surgeons make decisions informed by the most current and locally relevant data available.
This is where design, engineering, and clinical insight must converge.
At Kaleidoscope Innovation, together with our parent company Infosys, we work at the intersection of the physical and digital healthcare ecosystem—helping medtech organizations bring these next-generation capabilities, many of them AI-revolutionized, to life.
In addition to design, workflow and human factors insight; advanced engineering and digital system architecture, and regulatory and medical affairs expertise, Kaleidoscope’s AI-related work centers around:
- Dataset evaluation
- Model training & validation
- Usability (UX) & integration within the surgical system
While Infosys focuses on:
- Ethical AI model creation
- Cyber Security considerations
The highest order of value we offer clients is an end-to-end development capability that transforms complex data ecosystems into clinically meaningful, regulatory-ready solutions.
The future of surgical planning will not come from AI alone. It will come from thoughtfully designed systems that turn real-world clinical data into real-time clinical wisdom.
If you're exploring how AI-enabled platforms, smart devices, or connected surgical ecosystems might fit into your roadmap, I'd welcome the conversation.
Back to Insights + News